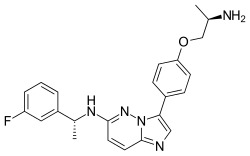
Taletrectinib
 | |
| Clinical data | |
|---|---|
| Trade names | Ibtrozi |
| AHFS/Drugs.com | Ibtrozi |
| License data | |
| Routes of administration | By mouth |
| Drug class | Antineoplastic |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL |
|
| Chemical and physical data | |
| Formula | C23H24FN5O |
| Molar mass | 405.477 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Taletrectinib, sold under the brand name Ibtrozi, is an anti-cancer medication used for the treatment of non-small cell lung cancer.[1][2] It is used as the salt, taletrectinib adipate.[1] Taletrectinib is a kinase inhibitor.[1] It is taken by mouth.[1]
Taletrectinib was approved for medical use in the United States in June 2025.[3]
Medical uses
Taletrectinib is indicated for the treatment of adults with locally advanced or metastatic ROS1-positive non-small cell lung cancer.[1][2]
Adverse effects
The FDA prescribing information for taletrectinib includes warnings and precautions for hepatotoxicity, interstitial lung disease/pneumonitis, QTc interval prolongation, hyperuricemia, myalgia with creatine phosphokinase elevation, skeletal fractures, and embryo-fetal toxicity.[1][3]
History
The efficacy of taletrectinib to treat ROS1-positive non-small cell lung cancer was evaluated in participants with locally advanced or metastatic, ROS1-positive non-small cell lung cancer enrolled in two multi-center, single-arm, open-label clinical trials, TRUST-I (NCT04395677) and TRUST-II (NCT04919811).[3] The efficacy population included 157 participants (103 in TRUST-I; 54 in TRUST-II) who were naïve to treatment with a ROS1 tyrosine kinase inhibitor (TKI) and 113 participants (66 in TRUST-I; 47 in TRUST-II) who had received one prior ROS1 tyrosine kinase inhibitor.[3] Participants may have received prior chemotherapy for advanced disease.[3] The US Food and Drug Administration (FDA) granted the application for taletrectinib priority review, breakthrough therapy, and orphan drug designations.[3]
Society and culture
Legal status
Taletrectinib was approved for medical use in the United States in June 2025.[3][4]
Names
Taletrectinib is the international nonproprietary name.[5]
Taletrectinib is sold under the brand name Ibtrozi.[3][4]
References
- ^ a b c d e f g "Ibtrozi- taletrectinib capsule". DailyMed. 11 June 2025. Retrieved 6 July 2025.
- ^ a b Khan I, Sahar A, Numra S, Saha N, Nidhi, Parveen R (April 2025). "Efficacy and safety of taletrectinib for treatment of ROS1 positive non-small cell lung cancer: A systematic review". Expert Opinion on Pharmacotherapy. 26 (6): 765–772. doi:10.1080/14656566.2025.2487150. PMID 40170301.
- ^ a b c d e f g h "FDA approves taletrectinib for ROS1-positive non-small cell lung cancer". U.S. Food and Drug Administration (FDA). 11 June 2025. Retrieved 13 June 2025.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "U.S. Food and Drug Administration Approves Nuvation Bio's Ibtrozi (taletrectinib), a Next-Generation Oral Treatment for Advanced ROS1-Positive Non-Small Cell Lung Cancer". Nuvation Bio (Press release). 12 June 2025. Retrieved 13 June 2025.
- ^ World Health Organization (2021). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 85". WHO Drug Information. 35 (1). hdl:10665/340684.
External links
- Clinical trial number NCT04395677 for "A Study of AB-106 in Subjects With Advanced NSCLC Harboring ROS1 Fusion Gene" at ClinicalTrials.gov
- Clinical trial number NCT04919811 for "Taletrectinib Phase 2 Global Study in ROS1 Positive NSCLC (TRUST-II)" at ClinicalTrials.gov